Medicine:Fertility medication
Fertility medications, also known as fertility drugs, are medications which enhance reproductive fertility. For women, fertility medication is used to stimulate follicle development of the ovary.[1] There are very few fertility medication options available for men.[2]
Agents that enhance ovarian activity can be classified as either gonadotropin releasing hormone, estrogen antagonists or gonadotropins.
Treatment decision-making involves four major factors: efficacy, burden of treatment (such as frequency of injections and office visits), safety, and financial costs.[3]
Female
Main techniques
The main techniques involving fertility medication in females are:
- Ovulation induction, with the aim of producing one or two ovulatory follicles for fertilization by sexual intercourse or artificial insemination
- Controlled ovarian hyperstimulation, which is generally part of in vitro fertilization, and the aim is generally to develop multiple follicles (optimally between 11 and 14 antral follicles measuring 2–8 mm in diameter), followed by transvaginal oocyte retrieval, co-incubation, followed by embryo transfer of a maximum of two embryos at a time.[4]
- Final maturation induction of follicles, also triggering a predictable time of ovulation.
Gonadotropin-releasing hormone
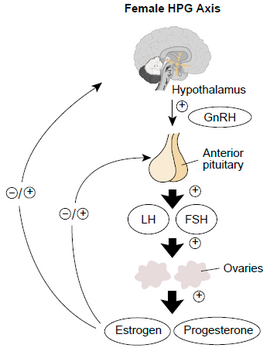
Either gonadotropin-releasing hormone (GnRH) or any gonadotropin-releasing hormone agonist (such as Lupron) may be used in combination with luteinizing hormone (LH) using an infusion pump to simulate endogenous hormone production. GnRH stimulates the release of gonadotropins (LH and FSH) from the anterior pituitary in the body. This set of therapy is reserved for a subset of women with infertility and have produced ovulation rates of 90% and pregnancy rates of 80% or higher.
Antiestrogens
Antiestrogens inhibit the effects of estrogen, which include selective estrogen receptor modulators (SERM) and aromatase inhibitors.
Selective estrogen receptor modulators
Clomiphene is a selective estrogen receptor modulator (SERM).[5] It is the most widely used fertility drug.[6] Other medications in this class include tamoxifen and raloxifene, although both are not as effective as clomiphene and are thus less widely used for fertility purposes.[7] They are used in ovulation induction by inhibiting the negative feedback of estrogen at the hypothalamus. As the negative feedback of estrogen is inhibited, the hypothalamus secretes GnRh which in turn stimulates the anterior pituitary to secrete LH and FSH which help in ovulation. Between 60 and 85% of women, mostly with polycystic ovary syndrome (PCOS), ovulate successfully in response to clomiphene with a cumulative pregnancy rate of 30 to 40%.[8][9][10]
Aromatase inhibitors
Although primarily used in breast cancer treatment, aromatase inhibitors (particularly generic letrozole) are also used in ovulation induction. Aromatase inhibitors are a common fertility treatment to treat women with PCOS. A meta-analysis analyzing live birth rates for women with PCOS treated with clomiphene compared to letrozole found that letrozole resulted in higher live birth rates.[11] However, ovulation induction remains an off-label indication, which affects use.
Gonadotropins
Gonadotropins are protein hormones that stimulate the gonads (testes and ovaries). For medication, they can be extracted from urine in postmenopausal women or through genetic modification and bacterial recombination. Examples of recombinant FSH are Follistim and Gonal F, while Luveris is a recombinant LH. FSH and recombinant FSH analogues are mainly used for controlled ovarian hyperstimulation as well as ovulation induction.[12] There has been some controversy over the efficacy between extracted and recombinant FSH for ovulation induction; however, a meta-analysis of 14 trials among 1726 women found that there were no differences in clinical pregnancy or live birth outcomes.[13]
Chemotherapy treatment in premenopausal women can compromise ovarian reserve and function, with gonadotoxic effects ranging from temporary to permanent infertility and premature ovarian failure (POF). Proposed mechanisms for chemotherapy-induced ovarian damage include apoptosis of growing follicles, fibrosis of stromal cells, and injury to blood vessels resulting in ischemia. First-line options for fertility preservation include embryo and oocyte preservation before starting chemotherapy, though these methods do not contribute to the preservation of gonadal function. GnRH agonist therapies have been associated with relatively low risk, time, and cost.[14] There is evidence that chemotherapy cotreatment with gonadotropin-releasing hormone (GnRH) can increase the probability of spontaneous menses and ovulation resumption. However, this cotreatment has not shown an improvement in pregnancy rates.[15]
Human chorionic gonadotropin
Human chorionic gonadotropin (hCG), also known as the “hormone of pregnancy” is a hormone that is normally produced during pregnancy and plays an integral role throughout reproduction.[16] It is crucial in maintaining pregnancy, from the stages of placentation to early embryo development.[16] It is also used in assisted reproductive techniques as it can be used to replace LH in final maturation induction.[16]
Other medications
Although metformin has been used off-label to treat oligomenorrhea and ovarian hyperstimulation syndrome (OHSS) in women with PCOS, metformin is no longer recommended as infertility treatment per the American Society for Reproductive Medicine (ASRM) in 2017. Its use to treat anovulatory infertility was based on an association of insulin resistance in non-obese women with PCOS. While metformin may increase ovulation in women with PCOS, there is no evidence of increased pregnancy rates or live-birth rates, and the combination therapy of metformin and clomiphene citrate did not provide a significant benefit compared to clomiphene citrate alone. First-line therapy for ovulation induction in women with PCOS remains the anti-estrogen clomiphene citrate or the aromatase inhibitor letrozole.[17]
Male
Treatment for oligospermia is centered around underlying causes, such as endocrine and systematic disorders that can cause hypogonadism.[18]
Typically, other assisted reproductive technologies are used. Although there is no FDA indication for use of aromatase inhibitors improving spermatogenesis, testolactone has been shown to be effective when compared to placebo.[19]
Though there is no FDA indication for the use of clomiphene in male infertility, it has been prescribed since the 1960s.[20] As of 2013, there is not substantial evidence to suggest that clomiphene can treat male infertility.[20]
Combinations of vitamins and minerals, including selenium, co-enzyme Q10, L-carnitine, folic acid, zinc, eicosapentaenoic acid (EPA), and docosahexaenoic acid (DHA), have been shown to improve male infertility, but due to the low amounts of studies and participants, more clinical studies are needed.[21] Folate in combination with zinc supplementation was shown to have a statistically significant effect on sperm concentration and morphology when compared to placebo.[22] There is evidence suggesting a significant association between vitamin D serum concentrations and the quality of sperm in men, characterized by the sperm's motility and progress motility.[23] Because the quality of men's sperm is influenced by genetics, results should be interpreted cautiously. There is little evidence that supplementation with antioxidants, such as pentoxifylline will increase male fertility.[24][25]
As of September 2017, mesenchymal stem cell therapy for infertility has been studied in animals, but has not entered clinical trials.[26] Stem cells collected from bone marrow and umbilical cord have shown the most ability to rehabilitate fertility in animals, but more studies are needed to determine efficacy.[26]
Adverse effects
Cancer
Since infertility increases the risk of ovarian cancer, fertility drugs have been used to combat this but the cancer risks are still not completely known.[27] (As of 2019) there have been studies that have shown the risk of developing ovarian cancer is higher when taking fertility medications. However, due to the low number of studies, lack of follow-up time and other contribution factors, the risk is unclear.[27] Most studies conducted have shown that fertility drugs do not increase the risk of other gynecologic cancers (cervical and endometrial) or other malignant cancers (thyroid, colon, melanoma, breast).[28] The validity of these data may be affected by patient-reported biases, small subject numbers, and other confounding variables.[28]
Children born to mothers who use fertility medication to induce ovulation are more than twice as likely to develop leukemia during their childhoods than other children.[29]
Ovarian hyperstimulation syndrome
Estrogen antagonists and gonadotropins may stimulate multiple follicles and other ovarian hormones leading to multiple birth and possible ovarian hyperstimulation syndrome (OHSS).[30] Development of OHSS is dependent on the administration of hCG and is mediated through vascular endothelial growth factor (VEGF). OHSS is characterized as cystic enlargement of the ovaries. Multiple birth is especially deleterious due to compounding risks including premature delivery and low birthweight, pre-eclampisa, and increased risk of neonatal mortality. While triplet births have been declining in ART, multiple births remain over 50% of births from IVF. However, there are limitations to measure, as 4% to 8% IVF clinics to do not report their data to the CDC.
Discontinuation
Main reasons for discontinuation across all types of fertility treatment and treatment stage, are "postponement of treatment, physical and psychological burden and relational and personal problems".[31]
See also
References
- ↑ "Patient Education: Infertility in Women". UpToDate. Waltham, MA: UpToDate. 2020.
- ↑ Impacts of Medications on Male Fertility. Cham, Switzerland: Palgrave Macmillan. 2017. ISBN 978-3-319-69535-8. https://www.springer.com/gp/book/9783319695341. Retrieved 23 February 2019.
- ↑ ""Patient-centered fertility treatment": what is required?". Fertility and Sterility 101 (4): 924–6. April 2014. doi:10.1016/j.fertnstert.2013.12.045. PMID 24502889.
- ↑ "Fertility problems: assessment and treatment". NICE clinical guideline CG156. U.K. National Institute for Health and Care Excellence (NICE). February 2013. http://guidance.nice.org.uk/CG156.
- ↑ "Selective estrogen receptor modulators (SERM)/Selektive Ostrogenrezeptormodulatoren (SERM).". Gynakologische Endokrinologie 13 (2): 126. 2015. doi:10.1007/s10304-015-0003-9. http://doc.rero.ch/record/332590/files/10304_2015_Article_3.pdf.
- ↑ "Reproductive endocrinology: revisiting ovulation induction in PCOS". Nature Reviews. Endocrinology 10 (12): 704–5. December 2014. doi:10.1038/nrendo.2014.156. PMID 25178733.
- ↑ "A pharmacological review of selective oestrogen receptor modulators". Human Reproduction Update 6 (3): 212–24. 2000. doi:10.1093/humupd/6.3.212. PMID 10874566.
- ↑ "Development, pharmacology and clinical experience with clomiphene citrate". Human Reproduction Update 2 (6): 483–506. 1996. doi:10.1093/humupd/2.6.483. PMID 9111183.
- ↑ "Ovulation and pregnancy rates with clomiphene citrate". Obstetrics and Gynecology 51 (3): 265–9. March 1978. doi:10.1097/00006250-197803000-00002. PMID 628527.
- ↑ "A decade's experience with an individualized clomiphene treatment regimen including its effect on the postcoital test". Fertility and Sterility 37 (2): 161–7. February 1982. doi:10.1016/s0015-0282(16)46033-4. PMID 7060766.
- ↑ Franik, Sebastian; Le, Quang-Khoi; Kremer, Jan Am; Kiesel, Ludwig; Farquhar, Cindy (2022-09-27). "Aromatase inhibitors (letrozole) for ovulation induction in infertile women with polycystic ovary syndrome". The Cochrane Database of Systematic Reviews 2022 (9): CD010287. doi:10.1002/14651858.CD010287.pub4. ISSN 1469-493X. PMID 36165742.
- ↑ "The role of recombinant LH in women with hypo-response to controlled ovarian stimulation: a systematic review and meta-analysis". Reproductive Biology and Endocrinology 17 (1): 18. February 2019. doi:10.1186/s12958-019-0460-4. PMID 30728019.
- ↑ "Gonadotrophins for ovulation induction in women with polycystic ovary syndrome". The Cochrane Database of Systematic Reviews 1 (1): CD010290. January 2019. doi:10.1002/14651858.CD010290.pub3. PMID 30648738.
- ↑ "Preservation of gonadal function in women undergoing chemotherapy: a systematic review and meta-analysis of the potential role for gonadotropin-releasing hormone agonists". Journal of Assisted Reproduction and Genetics 35 (4): 571–581. April 2018. doi:10.1007/s10815-018-1128-2. PMID 29470701.
- ↑ "Gonadotropin-releasing hormone analog cotreatment for preservation of ovarian function during gonadotoxic chemotherapy: a systematic review and meta-analysis". Fertility and Sterility 95 (3): 906–14.e1–4. March 2011. doi:10.1016/j.fertnstert.2010.11.017. PMID 21145541.
- ↑ 16.0 16.1 16.2 "Gonadotropin therapy in assisted reproduction: an evolutionary perspective from biologics to biotech". Clinics 69 (4): 279–93. 2014. doi:10.6061/clinics/2014(04)10. PMID 24714837.
- ↑ "Role of metformin for ovulation induction in infertile patients with polycystic ovary syndrome (PCOS): a guideline". Fertility and Sterility 108 (3): 426–441. September 2017. doi:10.1016/j.fertnstert.2017.06.026. PMID 28865539.
- ↑ "Male infertility". Lancet 349 (9054): 787–90. March 1997. doi:10.1016/s0140-6736(96)08341-9. PMID 9074589.
- ↑ "Aromatase inhibitors in the treatment of oligozoospermic or azoospermic men: a systematic review of randomized controlled trials". JBRA Assisted Reproduction 20 (2): 82–8. May 2016. doi:10.5935/1518-0557.20160019. PMID 27244767.
- ↑ 20.0 20.1 "Clomiphene for the treatment of male infertility". Reproductive Sciences 20 (7): 739–44. July 2013. doi:10.1177/1933719112466304. PMID 23202725.
- ↑ "Influence of oral vitamin and mineral supplementation on male infertility: a meta-analysis and systematic review". Reproductive Biomedicine Online 39 (2): 269–279. August 2019. doi:10.1016/j.rbmo.2019.03.099. PMID 31160241.
- ↑ "The Effect of Folate and Folate Plus Zinc Supplementation on Endocrine Parameters and Sperm Characteristics in Sub-Fertile Men: A Systematic Review and Meta-Analysis". Urology Journal 14 (5): 4069–4078. August 2017. PMID 28853101.
- ↑ "The association between serum vitamin D, fertility and semen quality: A systematic review and meta-analysis". International Journal of Surgery 71: 101–109. November 2019. doi:10.1016/j.ijsu.2019.09.025. PMID 31561004.
- ↑ "Antioxidants in fertility: impact on male and female reproductive outcomes". Fertility and Sterility 110 (4): 578–580. September 2018. doi:10.1016/j.fertnstert.2018.05.028. PMID 30196940.
- ↑ "On-label and off-label drugs used in the treatment of male infertility". Fertility and Sterility 103 (3): 595–604. March 2015. doi:10.1016/j.fertnstert.2014.12.122. PMID 25660648.
- ↑ 26.0 26.1 "Mesenchymal Stem Cells (MSCs) Therapy for Recovery of Fertility: a Systematic Review". Stem Cell Reviews and Reports 14 (1): 1–12. February 2018. doi:10.1007/s12015-017-9765-x. PMID 28884412.
- ↑ 27.0 27.1 "Risk of ovarian cancer in women treated with ovarian stimulating drugs for infertility". The Cochrane Database of Systematic Reviews 2019 (6): CD008215. June 2019. doi:10.1002/14651858.cd008215.pub3. PMID 31207666.
- ↑ 28.0 28.1 "Use of fertility medications and cancer risk: a review and update". Current Opinion in Obstetrics & Gynecology 29 (4): 195–201. August 2017. doi:10.1097/GCO.0000000000000370. PMID 28538003.
- ↑ "Fertility treatments, congenital malformations, fetal loss, and childhood acute leukemia: the ESCALE study (SFCE)". Pediatric Blood & Cancer 60 (2): 301–308. February 2013. doi:10.1002/pbc.24192. PMID 22610722.
- ↑ "Revisiting ovarian hyper stimulation syndrome: Towards OHSS free clinic". J Hum Reprod Sci 8 (1): 13–7. 2015. doi:10.4103/0974-1208.153120. PMID 25838743.
- ↑ "Why do patients discontinue fertility treatment? A systematic review of reasons and predictors of discontinuation in fertility treatment". Human Reproduction Update 18 (6): 652–69. August 2012. doi:10.1093/humupd/dms031. PMID 22869759.
 |

