Chemistry:Potassium permanganate
| |
| |

| |
| Names | |
|---|---|
| IUPAC name
Potassium manganate(VII)
| |
| Systematic IUPAC name
Potassium permanganate | |
| Other names
Chameleon mineral
Condy's crystals Permanganate of potash Hypermangan Purple potion powder | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 1490 |
| |
| |
| Properties | |
| KMnO4 | |
| Molar mass | 158.034 g/mol |
| Appearance | Purplish-bronze-gray needles purple in solution[1] |
| Odor | odorless |
| Melting point | 240 °C (464 °F; 513 K) (decomposes) |
| 76 g/L (25 °C)[2] 250 g/L (65 °C) | |
| Solubility | decomposes in alcohol and organic solvents |
| +20.0·10−6 cm3/mol[2]:4.134 | |
Refractive index (nD)
|
1.59 |
| Structure[3] | |
| Orthorhombic, oP24 | |
| Pnma, No. 62 | |
a = 0.909 nm, b = 0.572 nm, c = 0.741 nm
| |
Formula units (Z)
|
4 |
| Thermochemistry | |
Heat capacity (C)
|
119.2 J/mol K |
Std molar
entropy (S |
171.7 J K−1 mol−1 |
Std enthalpy of
formation (ΔfH⦵298) |
−813.4 kJ/mol |
Gibbs free energy (ΔfG˚)
|
-713.8 kJ/mol |
| Pharmacology | |
| 1=ATC code }} | D08AX06 (WHO) V03AB18 (WHO) |
| Hazards | |
| GHS pictograms |   
|
| NFPA 704 (fire diamond) | |
| Related compounds | |
Other anions
|
Potassium pertechnetate Potassium perrhenate |
Other cations
|
Sodium permanganate Ammonium permanganate Calcium permanganate Silver permanganate |
Related manganates
|
Potassium hypomanganate Potassium manganate |
Related compounds
|
Manganese heptoxide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
| Clinical data | |
|---|---|
| License data | |
Potassium permanganate is an inorganic compound with the chemical formula KMnO4. It is a purplish-black crystalline salt, that dissolves in water as K+ and MnO−4, an intensely pink to purple solution.
Potassium permanganate is widely used in the chemical industry and laboratories as a strong oxidizing agent, and also as a medication for dermatitis, for cleaning wounds, and general disinfection. It is on the World Health Organization's List of Essential Medicines.[4] In 2000, worldwide production was estimated at 30,000 tons.[4]
Properties
Potassium permanganate is the potassium salt of the tetrahedral transition metal oxo complex permanganate, in which four O2− ligands are bound to a manganese(VII) center.[citation needed]
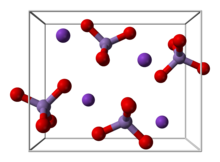
Structure
KMnO
4 forms orthorhombic crystals with constants: a = 910.5 pm, b = 572.0 pm, c = 742.5 pm. The overall motif is similar to that for barium sulfate, with which it forms solid solutions.[5] In the solid (as in solution), each MnO−
4 centre is tetrahedral. The Mn–O distances are 1.62 Å.[6]
Color
The purplish-black color of solid potassium permanganate, and the intensely pink to purple color of its solutions, is caused by its permanganate anion, which gets its color from a strong charge-transfer absorption band caused by excitation of electrons from oxo ligand orbitals to empty orbitals of the manganese(VII) center.[7]
Medical uses
Mechanism of action
Potassium permanganate functions as a strong oxidising agent.[8] Through this mechanism it results in disinfection, astringent effects, and decreased smell.[8]
Clinical use
Potassium permanganate is used for a number of skin conditions.[9] This includes fungal infections of the foot, impetigo, pemphigus, superficial wounds, dermatitis, and topical ulcers.[10][9] For topical ulcers it is used together with procaine benzylpenicillin. [9] Typically it is used in skin conditions that produce a lot of liquid.[10] It can be applied as a soaked dressing or a bath. It is on the World Health Organization's List of Essential Medicines.[4]
It can be used in children and adults.[11] It can be applied as a soaked dressing or a bath.[12] Petroleum jelly may be used on the nails before soaking to prevent their discoloration.[13] For treating eczema, it is recommended using for a few days at a time due to the possibility of it irritating the skin.[14]
The U.S. Food and Drug Administration does not recommend its use in the crystal or tablet form. It should only be used in a diluted liquid form.[15]
Historical use
Potassium permanganate was first made in the 1600s and came into common medical use at least as early as the 1800s.[16] During World War I Canadian soldiers were given potassium permanganate (to be applied mixed with an ointment) in an effort to prevent sexually transmitted infections (resulting mostly in violet stained genitals.)[17] Some have attempted to bring about an abortion by putting it in the vagina, though this is not effective.[18][19][20] Other historical uses have included as an effort to wash out the stomach in those with strychnine or picrotoxin poisoning.[21]
Side effects
Side effects from topical use may include irritation of the skin and discoloration of clothing.[22] A harsh burn on a child from an undissolved tablet has been reported.[23] Higher concentration solutions can result in chemical burns.[24] Therefore, the British National Formulary recommends 100 mg be dissolved in a liter of water before use to form a 1:10,000 (0.01%) solution.[25] [26][27] Wrapping the dressings soaked with potassium permanganate is not recommended.[citation needed]
Potassium permanganate is toxic if taken by mouth.[28] Side effects may include nausea, vomiting, and shortness of breath may occur.[29] If a sufficiently large amount (about 10 grams) is eaten death may occur.[29]
Concentrated solutions when drunk have resulted in Acute Respiratory Distress Syndrome or swelling of the airway.[30] Recommended measures for those who have ingested potassium permanganate include gastroscopy.[30] Activated charcoal or medications to cause vomiting are not recommended. While medications like ranitidine and N-acetylcysteine may be used in toxicity, evidence for this use is poor.[30]
Pharmaceuticals
In the United States the FDA requires tablets of the medication to be sold by prescription.[31] Potassium permanganate, however, does not have FDA approved uses and therefore non medical grade potassium permanganate is sometimes used for medical use.[citation needed]
It is available under a number of brand names including Permasol, Koi Med Tricho-Ex, and Kalii permanganas RFF.[32] It is occasionally called "Condy's crystals".[33]
Veterinary medicine
Potassium permanganate may be used to prevent the spread of glanders among horses.[34]
Industrial and other uses
Almost all applications of potassium permanganate exploit its oxidizing properties.[35] As a strong oxidant that does not generate toxic byproducts, KMnO4 has many niche uses.[citation needed]
Water treatment
Potassium permanganate is used extensively in the water treatment industry. It is used as a regeneration chemical to remove iron and hydrogen sulfide (rotten egg smell) from well water via a "manganese greensand" filter. "Pot-Perm" is also obtainable at pool supply stores and is used additionally to treat wastewater. Historically it was used to disinfect drinking water[36][37] and can turn the water pink.[38] Modern hiking and survivalist guides advise against using potassium permanganate in the field because it is difficult to dose correctly.[39] It currently finds application in the control of nuisance organisms such as zebra mussels in fresh water collection and treatment systems.[40]
Synthesis of organic compounds
A major application of KMnO4 is as a reagent for the synthesis of organic compounds.[41] Significant amounts are required for the synthesis of ascorbic acid, chloramphenicol, saccharin, isonicotinic acid, and pyrazinoic acid.[35]
KMnO4 is used in qualitative organic analysis to test for the presence of unsaturation. It is sometimes referred to as Baeyer's reagent after the German organic chemist Adolf von Baeyer . The reagent is an alkaline solution of potassium permanganate. Reaction with double or triple bonds (-C=C- or -C≡C-) causes the color to fade from purplish-pink to brown. Aldehydes and formic acid (and formates) also give a positive test.[42] The test is antiquated.
KMnO4 solution is a common thin layer chromatography stain for the detection of oxidizable functional groups, such as alcohols, aldehydes, alkenes, and ketones. Such compounds result in a white to orange spot on TLC plates.[43][44][45]
Analytical use
Potassium permanganate can be used to quantitatively determine the total oxidizable organic material in an aqueous sample. The value determined is known as the permanganate value. In analytical chemistry, a standardized aqueous solution of KMnO4 is sometimes used as an oxidizing titrant for redox titrations (permanganometry). As potassium permanganate is titrated, the solution becomes a light shade of purple, which darkens as excess of the titrant is added to the solution. In a related way, it is used as a reagent to determine the Kappa number of wood pulp. For the standardization of KMnO4 solutions, reduction by oxalic acid is often used.[46] In agricultural chemistry, it is used for estimation of active carbon in soil.[47]
Aqueous, acidic solutions of KMnO4 are used to collect gaseous mercury in flue gas during stationary source emissions testing.[48]
In histology, potassium permanganate was used as a bleaching agent.[49][50]
Fruit preservation
Ethylene absorbents extend storage time of bananas even at high temperatures. This effect can be exploited by packing bananas in polyethylene together with potassium permanganate. By removing ethylene by oxidation, the permanganate delays the ripening, increasing the fruit's shelf life up to 4 weeks without the need for refrigeration.[51][52][53]
Survival kits
Potassium permanganate is sometimes included in survival kits: as a hypergolic fire starter (when mixed with glycerol antifreeze from a car radiator);[54][55][56] as a water sterilizer; and for creating distress signals on snow.[57]
Fire service
Potassium permanganate is added to "plastic sphere dispensers" to create backfires, burnouts, and controlled burns. Polymer spheres resembling ping-pong balls containing small amounts of permanganate are injected with ethylene glycol and projected towards the area where ignition is desired, where they spontaneously ignite seconds later.[58][59] Both handheld[59] helicopter-[58] unmanned aircraft systems (UAS) or boat-mounted[59] plastic sphere dispensers are used.
Other uses
Potassium permanganate is one of the principal chemicals used in the film and television industries to "age" props and set dressings. Its ready conversion to brown MnO2 creates "hundred-year-old" or "ancient" looks on hessian cloth (burlap), ropes, timber, and glass.[60]
Potassium permanganate can be used to oxidize cocaine paste to purify it and increase its stability. This led to the Drug Enforcement Administration launching Operation Purple in 2000, with the goal of monitoring the world supply of potassium permanganate; however, potassium permanganate derivatives and substitutes were soon used thereafter to avoid the operation.[61]
Potassium permangate is used as an oxidizing agent in the synthesis of cocaine and methcathinone.[62]
Potassium permanganate is one of a number of possible treatments for Ichthyophthirius multifiliis (commonly known as "ich"), a parasite that infects and usually kills freshwater aquarium fish.
History
In 1659, Johann Rudolf Glauber fused a mixture of the mineral pyrolusite (manganese dioxide, MnO2) and potassium carbonate to obtain a material that, when dissolved in water, gave a green solution (potassium manganate) which slowly shifted to violet and then finally red.[63] The reaction that produced the color changes that Glauber observed in his solution of potassium permanganate and potassium manganate (K2MnO4) is now known as the "chemical chameleon". This report represents the first description of the production of potassium permanganate.[64] Just under 200 years later, London chemist Henry Bollmann Condy had an interest in disinfectants; he found that fusing pyrolusite with sodium hydroxide (NaOH) and dissolving it in water produced a solution with disinfectant properties. He patented this solution, and marketed it as 'Condy's Fluid'. Although effective, the solution was not very stable. This was overcome by using potassium hydroxide (KOH) rather than NaOH. This was more stable, and had the advantage of easy conversion to the equally effective potassium permanganate crystals. This crystalline material was known as 'Condy's crystals' or 'Condy's powder'. Potassium permanganate was comparatively easy to manufacture, so Condy was subsequently forced to spend considerable time in litigation to stop competitors from marketing similar products.[65]
Early photographers used it as a component of flash powder. It is now replaced with other oxidizers, due to the instability of permanganate mixtures.[citation needed]
Preparation
Potassium permanganate is produced industrially from manganese dioxide, which also occurs as the mineral pyrolusite. In 2000, worldwide production was estimated at 30,000 tonnes.[35] The MnO2 is fused with potassium hydroxide and heated in air or with another source of oxygen, like potassium nitrate or potassium chlorate.[35] This process gives potassium manganate:
- [math]\ce{ 2 MnO2 + 4 KOH + O2 -> 2 K2MnO4 + 2 H2O }[/math]
(With sodium hydroxide, the end product is not sodium manganate but an Mn(V) compound, which is one reason why the potassium permanganate is more commonly used than sodium permanganate. Furthermore, the potassium salt crystallizes better.[35])
The potassium manganate is then converted into permanganate by electrolytic oxidation in alkaline media:
- [math]\ce{ 2 K2MnO4 + 2 H2O -> 2 KMnO4 + 2 KOH + H2 }[/math]
Other methods
Although of no commercial importance, potassium manganate can be oxidized by chlorine or by disproportionation under acidic conditions.[66] The chlorine oxidation reaction is
- [math]\ce{ 2 K2MnO4 + Cl2 -> 2 KMnO4 + 2 KCl }[/math]
and the acid-induced disproportionation reaction may be written as
- [math]\ce{ 3 K2MnO4 + 4 HCl -> 2 KMnO4 + MnO2 + 2 H2O + 4 KCl }[/math]
A weak acid such as carbonic acid is sufficient for this reaction:
- [math]\ce{ 3 K2MnO4 + 2 CO2 -> 2 KMnO4 + 2 K2CO3 + MnO2 }[/math]
Permanganate salts may also be generated by treating a solution of Mn2+ ions with strong oxidants such as lead dioxide (PbO2), sodium bismuthate (NaBiO3), or peroxydisulfate. Tests for the presence of manganese exploit the vivid violet color of permanganate produced by these reagents.
Reactions
Organic chemistry
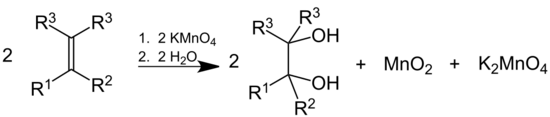
Dilute solutions of KMnO4 convert alkenes into diols. This behaviour is also used as a qualitative test for the presence of double or triple bonds in a molecule, since the reaction decolorizes the initially purple permanganate solution and generates a brown precipitate (MnO2). In this context, it is sometimes called Baeyer's reagent. However, bromine serves better in measuring unsaturation (double or triple bonds) quantitatively, since KMnO4, being a very strong oxidizing agent, can react with a variety of groups.
Under acidic conditions, the alkene double bond is cleaved to give the appropriate carboxylic acid:[67]
- [math]\ce{ CH3(CH2)17CH=CH2 + 2 KMnO4 + 3 H2SO4 -> CH3(CH2)17COOH + CO2 + 4 H2O + K2SO4 + 2 MnSO4 }[/math]
Potassium permanganate oxidizes aldehydes to carboxylic acids, illustrated by the conversion of n-heptanal to heptanoic acid:[68]
- [math]\ce{ 5 C6H13CHO + 2 KMnO4 + 3 H2SO4 -> 5 C6H13COOH + 3 H2O + K2SO4 + 2 MnSO4 }[/math]
Even an alkyl group (with a benzylic hydrogen) on an aromatic ring is oxidized, e.g. toluene to benzoic acid.[69]
- [math]\ce{ 5 C6H5CH3 + 6 KMnO4 + 9 H2SO4 -> 5 C6H5COOH + 14 H2O + 3 K2SO4 + 6 MnSO4 }[/math]
Glycols and polyols are highly reactive toward KMnO4. For example, addition of potassium permanganate to an aqueous solution of sugar and sodium hydroxide produces the chemical chameleon reaction, which involves dramatic color changes associated with the various oxidation states of manganese. A related vigorous reaction is exploited as a fire starter in survival kits. For example, a mixture of potassium permanganate and glycerol or pulverized glucose ignites readily.[54] Its sterilizing properties are another reason for inclusion of KMnO4 in a survival kit.[citation needed]
Ion exchange
Treating a mixture of aqueous potassium permanganate with a quaternary ammonium salt results in ion exchange, precipitating the quat salt of permanganate. Solutions of these salts are sometimes soluble in organic solvents:[70]
- [math]\ce{ KMnO4 + R4NCl -> R4NMnO4 + KCl }[/math]
Similarly, addition of a crown ether also gives a lipophilic salt.[71]
Reaction with acids and bases
Permanganate reacts with concentrated hydrochloric acid to give chlorine and manganese(II):
- [math]\ce{ 2 KMnO4 + 16 HCl -> 2 MnCl2 + 5 Cl2 + 2 KCl + 8 H2O }[/math]
In neutral solution, permanganate slowly reduces to manganese dioxide (MnO2). This is the material that stains one's skin when handling KMnO4.
KMnO4 reduces in alkaline solution to give green K2MnO4:[72]
- [math]\ce{ 4 KMnO4 + 4 KOH -> 4 K2MnO4 + O2 + 2 H2O }[/math]
This reaction illustrates the relatively rare role of hydroxide as a reducing agent.
Addition of concentrated sulfuric acid to potassium permanganate gives Mn2O7.[73] Although no reaction may be apparent, the vapor over the mixture will ignite paper impregnated with alcohol. Potassium permanganate and sulfuric acid react to produce some ozone, which has a high oxidizing power and rapidly oxidizes the alcohol, causing it to combust. As the reaction also produces explosive Mn2O7, this should only be attempted with great caution.[74][75]
Thermal decomposition
Solid potassium permanganate decomposes when heated:
- [math]\ce{ 2 KMnO4 -> K2MnO4 + MnO2 + O2 }[/math]
It is a redox reaction.
Safety and handling
Potassium permanganate poses risks as an oxidizer.[76] Contact with skin can cause skin irritation and in some cases severe allergic reaction. It can also result in discoloration and clothing stains.[77]
References
- ↑ (in Spanish) Química Analítica Cualitativa. Ediciones Paraninfo, S.A.. 1985. p. 688. ISBN 84-9732-140-5.
- ↑ 2.0 2.1 Cite error: Invalid
<ref>tag; no text was provided for refs namedcrc - ↑ "A stabilization factor in the polymorphism of ammonium nitrate" (in French). Comptes Rendus Hebdomadaires des Séances de l'Académie des Sciences 221: 261–263. 1945.
- ↑ 4.0 4.1 4.2 World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. 2019. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ Handbook of Preparative Inorganic Chemistry. New York: Academic Press. 1963.
- ↑ "Crystal structure of potassium permanganate". Inorg. Chem. 6 (3): 503–507. 1967. doi:10.1021/ic50049a015.
- ↑ Inorganic Chemistry (5th ed.). Pearson. 2014. p. 430. ISBN 978-0321811059.
- ↑ 8.0 8.1 "Potassium permanganate | DermNet New Zealand" (in en). https://www.dermnetnz.org/topics/potassium-permanganate/. Retrieved 11 October 2017.
- ↑ 9.0 9.1 9.2 WHO Model Formulary 2008. World Health Organization. 2009. pp. 295, 300. ISBN 9789241547659.
- ↑ 10.0 10.1 British Medical Association; Royal Pharmaceutical Society (2015). British national formulary (69 ed.). p. 840. ISBN 9780857111562.
- ↑ "WHO Model Prescribing Information: Drugs Used in Skin Diseases: Antiseptic agents: Potassium permanganate". http://apps.who.int/medicinedocs/en/d/Jh2918e/25.2.html. Retrieved 12 October 2017.
- ↑ WHO Model Formulary 2008. World Health Organization. 2009. pp. 295, 300. ISBN 9789241547659.
- ↑ "Medical Management" (in en). Oxford Handbook of Medical Dermatology. OUP Oxford. 2011. p. 592. ISBN 9780199558322. https://books.google.com/books?id=CDxsjyTSnawC&pg=PA592.
- ↑ "Should potassium permanganate be used in wound care?" (in en). 5 August 2003. https://www.nursingtimes.net/clinical-archive/wound-care/should-potassium-permanganate-be-used-in-wound-care/205220.article. Retrieved 12 October 2017.
- ↑ "CFR – Code of Federal Regulations Title 21". https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=250.108. Retrieved 11 October 2017.
- ↑ "Chapter 16" (in en). The Complete Idiot's Guide to Aquaponic Gardening. Penguin. 2013. p. 234. ISBN 9781615643332. https://books.google.com/books?id=BIfEfqS8iKcC&pg=RA1-PT234.
- ↑ "Pestilence and Mankind" (in en). A Short History of Medicine. Random House Publishing Group. 2008. p. 111. ISBN 9781588368218. https://books.google.com/books?id=bKpBZi8uG8IC&pg=PA111.
- ↑ "No Extras, Curbing Fertility During the Great Depression" (in en). Pregnancy and Power: A Short History of Reproductive Politics in America. NYU Press. 2005. p. 120. ISBN 9780814741191. https://books.google.com/books?id=Xe6gBwAAQBAJ&pg=PA120.
- ↑ (in en) Code of Federal Regulations: Record 2: 2007-. U.S. General Services Administration, National Archives and Records Service, Office of the Federal Register. 2008. p. 178. https://books.google.com/books?id=SbVRAQAAMAAJ.
- ↑ "CFR – Code of Federal Regulations Title 21". https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=250.108. Retrieved 11 October 2017.
- ↑ "Potassium permanganate definition | Drugs.com". https://www.drugs.com/dict/potassium-permanganate.html. Retrieved 11 October 2017.
- ↑ WHO Model Formulary 2008. World Health Organization. 2009. pp. 295, 300. ISBN 9789241547659.
- ↑ "Should potassium permanganate be used in wound care?" (in en). 5 August 2003. https://www.nursingtimes.net/clinical-archive/wound-care/should-potassium-permanganate-be-used-in-wound-care/205220.article. Retrieved 12 October 2017.
- ↑ (in en) Poisoning and Drug Overdose (Sixth ed.). McGraw Hill Professional. 2011. p. 121. ISBN 9780071716765. https://books.google.com/books?id=ju7diy73Z2wC.
- ↑ "Should potassium permanganate be used in wound care?" (in en). 5 August 2003. https://www.nursingtimes.net/clinical-archive/wound-care/should-potassium-permanganate-be-used-in-wound-care/205220.article. Retrieved 12 October 2017.
- ↑ British Medical Association; Royal Pharmaceutical Society (2015). British national formulary (69 ed.). p. 840. ISBN 9780857111562.
- ↑ "WHO Model Prescribing Information: Drugs Used in Skin Diseases: Antiseptic agents: Potassium permanganate". http://apps.who.int/medicinedocs/en/d/Jh2918e/25.2.html. Retrieved 12 October 2017.
- ↑ "Principles of treatment in pediatric dermatology" (in en). Pediatric Dermatology E-Book. Elsevier Health Sciences. 2011. p. 131. ISBN 978-0723436652. https://books.google.com/books?id=tAlGLYplkacC&pg=PA131.
- ↑ 29.0 29.1 "Oxidizers" (in en). A Comprehensive Guide to the Hazardous Properties of Chemical Substances. John Wiley & Sons. 2007. p. 710. ISBN 9780471714583. https://books.google.com/books?id=-CRRJBVv5d0C&pg=PA710.
- ↑ 30.0 30.1 30.2 "Potassium Chloride and Potassium Permanganate" (in en). Medical Toxicology. Lippincott Williams & Wilkins. 2004. pp. 904–905. ISBN 9780781728454. https://books.google.com/books?id=BfdighlyGiwC&pg=PA904.
- ↑ "CFR – Code of Federal Regulations Title 21". https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfcfr/CFRSearch.cfm?fr=250.108. Retrieved 11 October 2017.
- ↑ "Potassium Permanganate - Drugs.com". https://www.drugs.com/international/potassium-permanganate.html. Retrieved 11 October 2017.
- ↑ "Potassium permanganate" (in en). DermNet New Zealand. https://www.dermnetnz.org/topics/potassium-permanganate/. Retrieved 11 October 2017.
- ↑ "Bacterial Skin Diseases" (in en). Equine Dermatology – E-Book. Elsevier Health Sciences. 2010. p. 168. ISBN 978-1437709216. https://books.google.com/books?id=f0-v1vOq3x4C&pg=PA168.
- ↑ 35.0 35.1 35.2 35.3 35.4 "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. 2002. doi:10.1002/14356007.a16_123.
- ↑ Assembly of Life Sciences (U.S.). Safe Drinking Water Committee (1977). Drinking water and health, Volume 2. National Academies Press. p. 98. ISBN 978-0-309-02931-5. https://books.google.com/books?id=uVlQ_TUP-f0C&pg=PA98I. Retrieved 2016-09-13.
- ↑ "Red faces over pink water". The Northern Advocate. 28 February 2005. http://www.northernadvocate.co.nz/local/news/red-faces-over-pink-water/3625948/.
- ↑ "Onoway apologizes for 'alarming' pink tap water". CBC News. 7 March 2017. http://www.cbc.ca/news/canada/edmonton/onoway-water-chemical-pink-1.4013808.
- ↑ Johannes Vogel (2014) (in German). Trinkwasserversorgung in Extremsituationen. Paul Pietsch Verlage GmbH. ISBN 978-3613507845.
- ↑ "EPA Guidance Manual Alternative Disinfectants and Oxidants". U.S. Environmental Protection Agency. 13 October 2015. https://www.epa.gov/dwreginfo/stage-1-and-stage-2-disinfectants-and-disinfection-byproducts-rules.
- ↑ "The Classical Permanganate Ion: Still a Novel Oxidant in Organic Chemistry". Synthesis 1987 (2): 85–127. 1987. doi:10.1055/s-1987-27859.
- ↑ "Baeyer Test". Department of Chemistry and Biochemistry. Central Connecticut State University. 2013. http://www.chemistry.ccsu.edu/glagovich/teaching/316/qualanal/tests/baeyertest.html.
- ↑ Thin Layer Chromatography stains http://www.reachdevices.com/TLC_stains.html
- ↑ Jork, H., Funk, W., Fischer, W., Wimmer, H. (1990): Thin-Layer Chromatography: Reagents and Detection Methods, Volume 1a, VCH, Weinheim, ISBN:3-527-278834
- ↑ Jork, H., Funk, W., Fischer, W., Wimmer, H. (1994): Thin-Layer Chromatography: Reagents and Detection Methods, Volume 1b, VCH, Weinheim
- ↑ "Revising the Mechanism of the Permanganate/Oxalate Reaction". J. Phys. Chem. A 108 (50): 11026. 2004. doi:10.1021/jp047061u. Bibcode: 2004JPCA..10811026K.
- ↑ "Quick Reference Guide: Potassium Permanganate Test for Active Carbon". Victoria, Australia. https://vro.agriculture.vic.gov.au/dpi/vro/vrosite.nsf/pages/soilhealth_potassium-permanganate.
- ↑ Code of Federal Regulations(7-1-07) Edition, Title 40, Part 60, Appendix A-8, Method 29, Section 7.3.1
- ↑ "Amyloidosis-where are we now and where are we heading?". Archives of Pathology & Laboratory Medicine 134 (4): 545–551. April 2010. doi:10.5858/134.4.545. PMID 20367306.
- ↑ "Chemical typing of amyloid protein contained in formalin-fixed paraffin-embedded biopsy specimens". American Journal of Clinical Pathology 116 (1): 135–142. July 2001. doi:10.1309/TWBM-8L4E-VK22-FRH5. PMID 11447744.
- ↑ "Potassium Permanganate as an Ethylene Absorbent in Polyethylene Bags to Delay the Ripening of Bananas During Storage". Australian Journal of Experimental Agriculture and Animal Husbandry 10 (43): 237. 1970. doi:10.1071/EA9700237.
- ↑ "Transport of Bananas at Ambient Temperatures using Polyethylene Bags". Tropical Agriculture (Trinidad) 48: 163–165. 1971.
- ↑ "Effect of Temperature on the Storage Life of bananas Held in Polyethylene Bags with an Ethylene Absorbent". Tropical Agriculture (Trinidad) 51: 23–26. 1974.
- ↑ 54.0 54.1 "Fire by Chemical Reaction". PrimitiveWays. http://www.primitiveways.com/chemical_fire.html.
- ↑ "Potassium Permanganate". http://pssurvival.com/PS/Chemistry/Uses_of_Potassium_Permanganate_2010.pdf#page=1.
- ↑ "Making Fire with Potassium Permanganate and Glycerin". thesurvivalcache. 3 November 2012. http://thesurvivalcache.weebly.com/thesurvivalcaches-blog/making-fire-with-potassium-permanganate-and-glycerin.
- ↑ "Distress Signals". Evening Post CXXI (107): 5. 7 May 1936. http://paperspast.natlib.govt.nz/cgi-bin/paperspast?a=d&d=EP19360507.2.20.
- ↑ 58.0 58.1 "Aerial Ignition, Plastic Sphere Dispenser Description". Missoula Technology and Development Center. USDA Forest Service. http://www.fs.fed.us/eng/aerial_ign/plsphere/describe.htm.
- ↑ 59.0 59.1 59.2 "Wetland Warrior". Dirty Jobs. Season 6. Episode 2.
- ↑ "Victor DeLor contractor profile". PaintPRO 2 (1). February 2000. http://www.paintpro.net/Articles/PP201/PP201-Contractor_Profile.cfm. Retrieved 2009-11-12. "One of the techniques DeLor is known for among designers and clients is the special effects he creates with various chemical solutions. When applied to wood surfaces, these chemicals give a weathered appearance to new wood. ... To achieve the aesthetic on interior surfaces, DeLor often uses a mixture of water and potassium permanganate, a dry powder chemical.".
- ↑ "The Terrifying Substances People Put in Cocaine". Vice Media. March 10, 2014. https://www.vice.com/en_ca/article/vdpb4y/the-cash-is-in-the-cut.
- ↑ "[Analysis of potassium permanganate as addictive drug "precursor"]". Archiv Fur Kriminologie 211 (5–6): 160–165. May 2003. PMID 12872685.
- ↑ Glauber, Johann Rudolph, Prosperitas Germaniae (The prosperity of Germany), part 3 (Amsterdam, (Netherlands): Johann Jansson, 1659), pp. 93–94. From pp. 93–94: " … donec tandem Magnesiam istam nitro fixo permixtam, in crucibulo forti coctione a nitro reseratam vidi, unde elegans color purpureus provenit, massam hanc effusam in pulvere redegi, aqua calida extraxi, per filtrum liquorem transmisi. Tandem vero elegantissimum purpureum, igneumque liquorem accepi, qui fere singulis horis in frigore tantummodo consistens colorem permutavit, sic ut jam viridis, jam caerulei, jam sanguinei coloris sponte sua factus sit, mox iterum alios elegantissimos colores receperit." ( … until finally I saw [that] by mixing that magnesia [i.e., magnesia nigra, pyrolusite, the ore containing manganese dioxide ] with fixed niter [i.e., inert niter, potassium carbonate], by cooking [it] in a strong crucible, [the colored compound was] released by the niter, whence a fine purple color arises; this mass [was] poured out, reduced to powder, extracted with hot water, [and] the solution passed through a filter. Then I got a truly most elegant, purple, and fiery solution, which nearly every hour ([while] standing just in the cold) changed color, so that it was spontaneously made now green, now blue, now red in color; soon again it received other most elegant colors.) "Prosperitatis Germaniae pars .... 3: In qua Salpetrae ex variis ubiq, obviis subiectis facillime atque copiose extrahendi modus traditur. Eiusve utilitates summae declarantur" (in Latin). https://www.digitale-sammlungen.de/en/view/bsb10915847?page=100. Translated in: The Works of the Highly Experienced and Famous Chymist, John Rudolph Glauber. London, England: Thomas Millbourn. 1689. p. 353. https://archive.org/stream/worksofhighlyexp00glau#page/352/mode/2up.
- ↑ Discovery of the Elements (6th ed.). Easton, Pennsylvania: Journal of Chemical Education. 1956. pp. 172–173. https://archive.org/stream/discoveryoftheel002045mbp#page/n185/mode/2up.
- ↑ "Important Trade Mark Case". Otago Witness 2 (2420): 53. 2 August 1900. http://www.paperspast.natlib.govt.nz/cgi-bin/paperspast?a=d&d=OW19000802.2.357.
- ↑ Inorganic Preparations. New York: Prentice-Hall. 1948. pp. 150–151.
- ↑ "Carboxylic Acids from the Oxidation of Terminal Alkenes by Permanganate: Nonadecanoic Acid". Organic Syntheses. 1990. http://www.orgsyn.org/demo.aspx?prep=cv7p0397.; Collective Volume, 7, pp. 397
- ↑ "n-Heptanoic acid". Organic Syntheses. http://www.orgsyn.org/demo.aspx?prep=cv2p0315.; Collective Volume, 2, pp. 315
- ↑ "Understanding C-H bond oxidations: H. and H- transfer in the oxidation of toluene by permanganate". Science 269 (5232): 1849–1851. September 1995. doi:10.1126/science.7569922. PMID 7569922. Bibcode: 1995Sci...269.1849G.
- ↑ "Purple benzene: Solubilization of anions in organic solvents". J. Chem. Educ. 54 (4): 229. 1977. doi:10.1021/ed054p229.1. Bibcode: 1977JChEd..54Q.229H.
- ↑ "Purple benzene revisited". J. Chem. Educ. 57 (4): 308. 1980. doi:10.1021/ed057p308.1. Bibcode: 1980JChEd..57..308D.
- ↑ "Manganates (VI)". Inorganic Syntheses. 11. 1968. pp. 56–61. doi:10.1002/9780470132425.ch11. ISBN 978-0-470-13242-5.
- ↑ Advanced Inorganic Chemistry (6th ed.). Wiley-VCH. 1999. ISBN 0-471-19957-5.
- ↑ "Clemens Winkler. His Experiments with Ozone in 1892". Praxis der Naturwissenschaften, Chemie 49: 18ff. 2000.
- ↑ "Formation of Ozone During the Reduction of Potassium Permanganate in Sulfuric Acid Solutions". Russian Journal of Physical Chemistry 79: 1755–1760. 2005.
- ↑ Bretherick's Handbook of Reactive Chemical Hazards. 1 (7th ed.). Elsevier Academic Press. 2007. pp. 1811–7. ISBN 978-0-12-373945-2.
- ↑ "How to Remove Potassium Permanganate". https://www.livestrong.com/article/74289-remove-potassium-permanganate/.
Further reading
- "Potassium permanganate". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/name/potassium%20permanganate.
- "Manganese and compounds Fact Sheet". National Pollutant Inventory. Australia: Department of Climate Change, Energy, the Environment and Water. http://www.npi.gov.au/resource/manganese-compounds.
- "The use of potassium permanganate in fish ponds". IFAS Extention. Gainesville, FL: University of Florida. 2009. http://edis.ifas.ufl.edu/fa032.
External links
 |