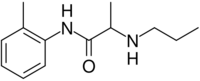
Chemistry:Prilocaine
 | |
| Clinical data | |
|---|---|
| Trade names | Citanest |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a603026 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Subcutaneous |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | 55% |
| Metabolism | Liver and kidney |
| Elimination half-life | 10-150 minutes, longer with impaired liver or kidney function |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C13H20N2O |
| Molar mass | 220.316 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
| Melting point | 37 to 38 °C (99 to 100 °F) |
| |
| |
| (verify) | |
Prilocaine (/ˈpraɪləˌkeɪn/[1]) is a local anesthetic of the amino amide type first prepared by Claes Tegner and Nils Löfgren. In its injectable form (trade name Citanest), it is often used in dentistry. It is also often combined with lidocaine as a topical preparation for dermal anesthesia (lidocaine/prilocaine or EMLA), for treatment of conditions like paresthesia. As it has low cardiac toxicity, it is commonly used for intravenous regional anaesthesia (IVRA).
Contraindications
In some patients, ortho-toluidine, a metabolite of prilocaine, may cause methemoglobinemia, which may be treated with methylene blue. Prilocaine may also be contraindicated in people with sickle cell anemia, anemia, or symptomatic hypoxia.[2]
Combinations
It is given as a combination with the vasoconstrictor epinephrine under the trade name Citanest Forte. It is used as a eutectic mixture with lidocaine, 50% w/w, as lidocaine/prilocaine. The mixture is an oil with a melting point of 18 °C (64 °F). A 5% emulsion preparation, containing 2.5% each of lidocaine/prilocaine, is marketed by APP Pharmaceuticals under the trade name EMLA (an abbreviation for eutectic mixture of local anesthetics).[3]
Compendial status
- United States Pharmacopeia 31 [4]
Synthesis
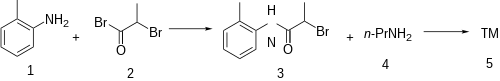
The amidation between o-toluidine [95-53-4] (1) and 2-bromopropionyl bromide [563-76-8] (2) leads to 2-bromo-N-(2-methylphenyl)propanamide [19397-79-6] (3). Displacement of the remaining halide with propylamine [107-10-8] (4) completed the synthesis of prilocaine (5).
See also
- Lidocaine/prilocaine
References
- ↑ "Prilocaine". Merriam-Webster Dictionary. https://www.merriam-webster.com/dictionary/Prilocaine. Retrieved 2016-01-21.
- ↑ Practical and Professional Clinical Skills. Oxford University Press. 2011-09-15. p. 267. ISBN 9780199585618. https://books.google.com/books?id=_DY5PCOFH4EC&pg=PA267.
- ↑ "Topical Anesthesia Use in Children: Eutectic Mixture of Local Anesthetics". Medscape.com. http://www.medscape.com/viewarticle/704761_2.
- ↑ The United States Pharmacopeial Convention, Revision Bulletin: Lidocaine and Prilocaine Cream–Revision to Related Compounds Test, http://www.usp.org/USPNF/notices/lidocaineAndPrilocaine.html, retrieved 10 July 2009
- ↑ Löfgren, Nils; Tegnér, Claës; Arwidsson, Barbro; Varde, E.; Westin, Gertrud (1960). "Studies on Local Anesthetics. XX. Synthesis of Some alpha-Monoalkylamino-2-methylpropionanilides. A New Useful Local Anesthetic.". Acta Chemica Scandinavica 14: 486–490. doi:10.3891/acta.chem.scand.14-0486.
- ↑ Anon., GB patent 839943 (to Astra Apotekarnes Kem Fab).
- ↑ Lofgren Nils Magnus, Tegner Claes Philip, U.S. Patent 3,160,662 (1964 to Astra Apotekarnes Kem Fab).
- ↑ Demare, Patricia; Regla, Ignacio (2012). "Synthesis of Two Local Anesthetics from Toluene: An Organic Multistep Synthesis in a Project-Oriented Laboratory Course". Journal of Chemical Education. 89 (1): 147–149. doi:10.1021/ed100838a.
- ↑ 沈文晖, 杨忠鑫, 葛小强, 张宇生, 邹玉龙, 张强, 杨继斌, 蔡中文, CN patent 105439887 (2016 to 重庆康乐制药有限公司).
External links
- "Prilocaine". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/name/prilocaine.
- "Prilocaine hydrochloride". Drug Information Portal. U.S. National Library of Medicine. https://druginfo.nlm.nih.gov/drugportal/name/prilocaine%20hydrochloride.
 |