Chemistry:Dabigatran
 | |
| Clinical data | |
|---|---|
| Trade names | Pradaxa, Pradax, Prazaxa, others |
| Other names | BIBR-953, BIBR-1048 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a610024 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth |
| Drug class | Direct thrombin inhibitor |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 3–7%[6] |
| Protein binding | 35%[6] |
| Elimination half-life | 12–17 hours[6] |
| Identifiers | |
| CAS Number |
|
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII |
|
| KEGG | |
| ChEBI |
|
| ChEMBL |
|
| PDB ligand | |
| Chemical and physical data | |
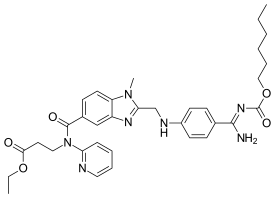
| Formula | C25H25N7O3 |
| Molar mass | 471.521 g·mol−1 |
| 3D model (JSmol) |
|
| |
| |
| | |
Dabigatran, sold under the brand name Pradaxa among others, is an anticoagulant used to treat and prevent blood clots and to prevent stroke in people with atrial fibrillation.[6][7] Specifically it is used to prevent blood clots following hip or knee replacement and in those with a history of prior clots.[6] It is used as an alternative to warfarin and does not require monitoring by blood tests.[6] In a meta analysis of 7 different studies, there was no benefit of dabigatran over warfarin in preventing ischemic stroke; however, dabigatran were associated with a lower hazard for intracranial bleeding compared with warfarin, but also had a higher risk of gastrointestinal bleeding relative to warfarin.[8] It is taken by mouth.[6]
Common side effects include bleeding and gastritis.[6] Other side effects may include bleeding around the spine and allergic reactions such as anaphylaxis.[6] In cases of severe bleeding, it can be reversed with the antidote, idarucizumab.[6] Use is not recommended during pregnancy or breastfeeding.[6] Compared to warfarin it has fewer interactions with other medications.[9] It is a direct thrombin inhibitor.[7]
Dabigatran was approved for medical use in the United States in 2010.[6] It is on the World Health Organization's List of Essential Medicines.[10] In 2020, it was the 306th most commonly prescribed medication in the United States, with more than 1 million prescriptions.[11] Dabigatran is available a generic medication.[12][13]
Medical uses
Dabigatran is used to prevent strokes in those with atrial fibrillation not caused by heart valve issues, as well as deep vein thrombosis and pulmonary embolism in persons who have been treated for 5–10 days with parenteral anticoagulant (usually low molecular weight heparin), and to prevent deep vein thrombosis and pulmonary embolism in some circumstances.[4]
It appears to be as effective as warfarin in preventing non-hemorrhagic strokes and embolic events in those with atrial fibrillation not due to valve problems.[14][15][16]
In 2022, a comparative study was performed on direct oral anticoagulants for patients with atrial fibrillation. Results demonstrated apixaban use was associated with lower risk for gastrointestinal bleeding and similar rates of ischemic stroke or systemic embolism, intracerebral brain hemorrhage, and all-cause mortality compared with dabigatran, edoxaban, and rivaroxaban.[17]
Contraindications
Dabigatran is contraindicated in patients who have active pathological bleeding, since dabigatran can increase bleeding risk and can also cause serious and potentially life-threatening bleeds.[4] Dabigatran is also contraindicated in patients who have a history of serious hypersensitivity reaction to dabigatran (e.g. anaphylaxis or anaphylactic shock).[4] The use of dabigatran should also be avoided in patients with mechanical prosthetic heart valves due to the increased risk of thromboembolic events (e.g. valve thrombosis, stroke, and myocardial infarction) and major bleeding associated with dabigatran in this population.[4][18][19]
Current FDA guidelines states that patients with mechanical heart valves should not be using dabigatran. The safety and efficacy of Pradaxa (dabigatran) were evaluated in the European RE-ALIGN trial in 2012. RE-ALIGN was terminated early because the Pradaxa treatment group had significantly more thromboembolic events and major bleeding than warfarin and determined to be contraindicated for use in patients with mechanical heart valves.[20] Further studies are needed in order to determine effects of dabigatran on patients with bioprosthetic valves.
Dabigatran is poorly excreted in breastmilk and does not appear to require any limitations to breastfeeding.[21] However, data is limited and further studies are needed.
Adverse effects
The most commonly reported side effect of dabigatran is gastrointestinal upset. When compared with people anticoagulated with warfarin, patients taking dabigatran had fewer life-threatening bleeds, fewer minor and major bleeds, including intracranial bleeds, but the rate of gastrointestinal bleeding was significantly higher. Dabigatran capsules contain tartaric acid, which lowers the gastric pH and is required for adequate absorption. The lower pH has previously been associated with dyspepsia; some hypothesize that this plays a role in the increased risk of gastrointestinal bleeding.[22] If a small amount of GI bleeding is diagnosed, the clinicians may consider adding H2 receptor inhibitor (H2RA), proton pump inhibitors (PPIs) and mucosal protective agent. In severe bleeding, measures include discontinuation of dabigatran immediately, and administration of prothrombin complex concentrate, packed red blood cells, fresh frozen plasma, the use of specific reversal agents such as idarucizumab for dabigatran, and emergency endoscopic management.[23]
A small but significantly increased risk of myocardial infarctions (heart attacks) has been noted when combining the safety outcome data from multiple trials.[24]
Reduced doses should be used in those with poor kidney function.[25]
Dabigatran intake has also been reported to cause esophageal injury or esophagitis. In a 2016 study by Toya et al., roughly 20% of patients suffered esophageal mucosa damage.[26] It has been theorized that the tartaric-acid core in the drug adheres and damages the esophagus, and then the damaged esophageal mucosa exfoliates after peristalsis.[27] Additionally, patients with limited mobility, reduced salivary secretion, and low water consumption will increase the possibility of contact by dabigatran with the esophageal mucosa.[23]
The Randomized Evaluation of Long-Term Anticoagulation Therapy (RE-LY) trial showed that impairment of liver function caused by dabigatran occurred in the same frequency as warfarin[28]
Pharmacology
Mechanism of action
Dabigatran reversibly binds to the active site on the thrombin molecule, preventing thrombin-mediated activation of coagulation factors. Furthermore, dabigatran can inactivate thrombin even when thrombin is fibrin-bound; it reduces thrombin-mediated inhibition of fibrinolysis and, therefore, may enhance fibrinolysis.[29]
Pharmacokinetics
Dabigatran has a half-life of approximately 12–14 h and exerts a maximum anticoagulation effect within 2–3 hours after ingestion.[citation needed] Fatty foods delay the intestinal absorption of dabigatran, although the bioavailability of the drug is unaffected.[4] One study showed that absorption may be moderately decreased if taken with a proton pump inhibitor.[30] Dabigatran excretion through P-glycoprotein pumps is slowed in patients taking strong p-glycoprotein pump inhibitors such as quinidine, verapamil, and amiodarone, thus raising plasma levels of dabigatran.[31]
Dabigatran is available as dabigatran etexilate mesilate, formulated as the prodrug dabigatran etexilate.[4][6][31]
History
Dabigatran (then compound BIBR-953) was discovered from a panel of chemicals with similar structure to benzamidine-based thrombin inhibitor α-NAPAP (N-alpha-(2-naphthylsulfonylglycyl)-4-amidinophenylalanine piperidide), which had been known since the 1980s as a powerful inhibitor of various serine proteases, specifically thrombin, but also trypsin. Addition of ethyl ester and hexyloxycarbonyl carbamide hydrophobic side chains led to the orally absorbed prodrug, BIBR 1048 (dabigatran etexilate).[32]
In March 2008, the European Medicines Agency (EMA) granted marketing authorization for Pradaxa for the prevention of thromboembolic disease following hip or knee replacement surgery and for non-valvular atrial fibrillation.[33]
The National Health Service (NHS) in Britain authorized dabigatran for use in preventing blood clots in hip and knee replacement surgery patients. According to a BBC article in 2008, Dabigatran was expected to cost the NHS £4.20 per day, which was similar to several other anticoagulants.[34]
Initially, there was no specific way to reverse the anticoagulant effect of dabigatran in the event of a major bleeding event,[35] unlike for warfarin.[36] Since then, the dabigatran-specific antidote idarucizumab, a humanized monoclonal antibody for intravenous administration, was developed, and received Food and Drug Administration (FDA) approval in 2015.[37]
Pradaxa received a Notice of Compliance (NOC) from Health Canada in June 2008,[38] for the prevention of blood clots in patients who have undergone total hip or total knee replacement surgery. Approval for atrial fibrillation patients at risk of stroke came in October 2010.[39][40]
The U.S. Food and Drug Administration (FDA) approved Pradaxa in October 2010, for prevention of stroke in patients with non-valvular atrial fibrillation.[41][42][43][44] The approval came after an advisory committee recommended the drug for approval in September 2010,[45] although caution is still urged by some outside experts.[46]
In February 2011, the American College of Cardiology Foundation and the American Heart Association added dabigatran to their guidelines for management of non-valvular atrial fibrillation with a class I recommendation.[47]
In May 2014, the FDA reported the results of a large study comparing dabigatran with warfarin in 134,000 Medicare patients. The agency concluded that dabigatran is associated with a lower risk of overall mortality, ischemic stroke, and bleeding in the brain than warfarin. Gastrointestinal bleeding was more common in those treated with dabigatran than in those treated with warfarin. The risk of heart attack was similar between the two drugs. The FDA reiterated its opinion that dabigatran's overall risk/benefit ratio is favorable.[48]
In July 2014, a series of investigations accused the privately held Boehringer Ingelheim pharmaceutical group of withholding critical information about the need for monitoring to protect patients from severe bleeding, particularly in the elderly. Review of internal communications between Boehringer researchers and employees by the FDA and the EMA revealed that Boehringer researchers had found evidence that serum levels of dabigatran vary widely. The BMJ investigation suggested that Boehringer had a financial motive to withhold this concern from regulatory health agencies because the data conflicted with their extensive marketing of dabigatran as an anticoagulant that does not require monitoring.[49][50] In August 2012, Pradaxa claims filed in U.S. federal courts were consolidated into a multi-district litigation in the Southern District of Illinois before Chief Judge David R. Herndon. In May 2014, a $650 million settlement was announced on behalf of approximately 3,900 claimants who were injured by the drug Pradaxa made by Boehringer Ingelheim Pharmaceuticals, Inc. The drug was alleged to cause severe bleeding events and/or hemorrhaging to those who were taking the drug.[51]
References
- ↑ "Dabigatran (Pradaxa) Use During Pregnancy". 27 December 2018. https://www.drugs.com/pregnancy/dabigatran.html.
- ↑ "Pradaxa Product information". 22 October 2009. https://health-products.canada.ca/dpd-bdpp/info?lang=eng&code=79794.
- ↑ "Summary of Product Characteristics (SmPC)". 15 September 2022. https://www.medicines.org.uk/emc/product/4703/smpc.
- ↑ 4.0 4.1 4.2 4.3 4.4 4.5 4.6 "Pradaxa- dabigatran etexilate mesylate capsule". 6 July 2020. https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=ba74e3cd-b06f-4145-b284-5fd6b84ff3c9.
- ↑ "Pradaxa EPAR". 22 March 2023. https://www.ema.europa.eu/en/medicines/human/EPAR/pradaxa.
- ↑ 6.00 6.01 6.02 6.03 6.04 6.05 6.06 6.07 6.08 6.09 6.10 6.11 6.12 "Dabigatran Etexilate Mesylate Monograph for Professionals". American Society of Health-System Pharmacists. https://www.drugs.com/monograph/dabigatran-etexilate-mesylate.html.
- ↑ 7.0 7.1 British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. pp. 135–137. ISBN 9780857113382.
- ↑ "Dabigatran Versus Warfarin for Atrial Fibrillation in Real-World Clinical Practice: A Systematic Review and Meta-Analysis". Circulation: Cardiovascular Quality and Outcomes 9 (2): 126–134. March 2016. doi:10.1161/CIRCOUTCOMES.115.002369. PMID 26812933.
- ↑ Oral Anticoagulation Therapy: Cases and Clinical Correlation. Springer. 2017. p. 11. ISBN 9783319546438. https://books.google.com/books?id=byYmDwAAQBAJ&pg=PA11.
- ↑ World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. 2019. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ↑ "Dabigatran - Drug Usage Statistics". https://clincalc.com/DrugStats/Drugs/Dabigatran.
- ↑ "Office of Generic Drugs 2020 Annual Report". https://www.fda.gov/drugs/generic-drugs/office-generic-drugs-2020-annual-report.
- ↑ "Dabigatran Etexilate Accord". 31 May 2023. https://www.ema.europa.eu/en/medicines/human/EPAR/dabigatran-etexilate-accord.
- ↑ "Dabigatran, Rivaroxaban, or Apixaban versus Warfarin in Patients with Nonvalvular Atrial Fibrillation: A Systematic Review and Meta-Analysis of Subgroups". Thrombosis 2013: 640723. 2013. doi:10.1155/2013/640723. PMID 24455237.
- ↑ "Interventions for Preventing Thromboembolic Events in Patients With Atrial Fibrillation: A Systematic Review". Annals of Internal Medicine 169 (11): 774–787. December 2018. doi:10.7326/M18-1523. PMID 30383133.
- ↑ "Stroke Prevention in Patients With Atrial Fibrillation: A Systematic Review Update". Agency for Healthcare Research and Quality (US) (Rockville (MD)). October 2018. doi:10.23970/ahrqepccer214. Report No.: 18-EHC018-EFReport No.: 2018-SR-04.. PMID 30480925. https://effectivehealthcare.ahrq.gov/topics/stroke-afib-update/research-2018. Retrieved 31 May 2023.
- ↑ "Comparative Effectiveness and Safety Between Apixaban, Dabigatran, Edoxaban, and Rivaroxaban Among Patients With Atrial Fibrillation : A Multinational Population-Based Cohort Study". Annals of Internal Medicine 175 (11): 1515–1524. November 2022. doi:10.7326/M22-0511. PMID 36315950.
- ↑ "FDA Drug Safety Communication: Pradaxa (dabigatran etexilate mesylate) should not be used in patients with mechanical prosthetic heart valves". https://www.fda.gov/Drugs/DrugSafety/ucm332912.htm.
- ↑ "Dabigatran versus warfarin in patients with mechanical heart valves". The New England Journal of Medicine 369 (13): 1206–1214. September 2013. doi:10.1056/NEJMoa1300615. PMID 23991661.
- ↑ "FDA Drug Safety Communication: Pradaxa (dabigatran etexilate mesylate) should not be used in patients with mechanical prosthetic heart valves" (in en). Center for Drug Evaluation and Research. U.S. Food and Drug Administration. 21 June 2019. https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-pradaxa-dabigatran-etexilate-mesylate-should-not-be-used-patients.
- ↑ "Dabigatran", Drugs and Lactation Database (LactMed®) (Bethesda (MD): National Institute of Child Health and Human Development), 2006, PMID 29999803, http://www.ncbi.nlm.nih.gov/books/NBK500744/, retrieved 27 February 2023
- ↑ "Dabigatran etexilate: A novel oral direct thrombin inhibitor". American Journal of Health-System Pharmacy 68 (16): 1506–1519. August 2011. doi:10.2146/ajhp100348. PMID 21817082.
- ↑ 23.0 23.1 "Dabigatran must be used carefully: literature review and recommendations for management of adverse events" (in English). Drug Design, Development and Therapy 13: 1527–1533. 6 May 2019. doi:10.2147/DDDT.S203112. PMID 31190734.
- ↑ "Dabigatran association with higher risk of acute coronary events: meta-analysis of noninferiority randomized controlled trials". Archives of Internal Medicine 172 (5): 397–402. March 2012. doi:10.1001/archinternmed.2011.1666. PMID 22231617.
- ↑ "18/12/2014 Pradaxa -EMEA/H/C/000829 -II/0073". http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_.[yes|permanent dead link|dead link}}]
- ↑ "Dabigatran-induced esophagitis: The prevalence and endoscopic characteristics". Journal of Gastroenterology and Hepatology 31 (3): 610–614. March 2016. doi:10.1111/jgh.13024. PMID 26102078.
- ↑ "Analysis of upper gastrointestinal adverse events among patients given dabigatran in the RE-LY trial". Clinical Gastroenterology and Hepatology 11 (3): 246–252.e5. March 2013. doi:10.1016/j.cgh.2012.10.021. PMID 23103906.
- ↑ "Dabigatran versus warfarin in patients with atrial fibrillation". The New England Journal of Medicine 361 (12): 1139–1151. September 2009. doi:10.1056/NEJMoa0905561. PMID 19717844.
- ↑ "Dabigatran (Pradaxa)". AJNR. American Journal of Neuroradiology 33 (3): 426–428. March 2012. doi:10.3174/ajnr.A3000. PMID 22345499.
- ↑ "Pharmacokinetic profile of the oral direct thrombin inhibitor dabigatran etexilate in healthy volunteers and patients undergoing total hip replacement". Journal of Clinical Pharmacology 45 (5): 555–563. May 2005. doi:10.1177/0091270005274550. PMID 15831779.
- ↑ 31.0 31.1 "Pradaxa Summary of Product Characteristics" (2018) . European Medicines Agency.
- ↑ "Structure-based design of novel potent nonpeptide thrombin inhibitors". Journal of Medicinal Chemistry 45 (9): 1757–1766. April 2002. doi:10.1021/jm0109513. PMID 11960487.
- ↑ "Pradaxa EPAR". European Medicines Agency. http://www.emea.europa.eu/ema/humandocs/Humans/EPAR/pradaxa/pradaxa.htm.
- ↑ "Clot drug 'could save thousands'". BBC News Online. 20 April 2008. http://news.bbc.co.uk/1/hi/health/7354818.stm.
- ↑ "Dabigatran etexilate--a novel, reversible, oral direct thrombin inhibitor: interpretation of coagulation assays and reversal of anticoagulant activity". Thrombosis and Haemostasis 103 (6): 1116–1127. June 2010. doi:10.1160/TH09-11-0758. PMID 20352166. "Although there is no specific antidote to antagonise the anticoagulant effect of dabigatran, due to its short duration of effect drug discontinuation is usually sufficient to reverse any excessive anticoagulant activity.".
- ↑ "Warfarin reversal". Journal of Clinical Pathology 57 (11): 1132–1139. November 2004. doi:10.1136/jcp.2003.008904. PMID 15509671.
- ↑ "Idarucizumab: A Review as a Reversal Agent for Dabigatran". American Journal of Cardiovascular Drugs 16 (4): 297–304. August 2016. doi:10.1007/s40256-016-0181-4. PMID 27388764.
- ↑ "Summary Basis of Decision (SBD): Pradax" Health Canada. 6 November 2008.
- ↑ "Approval of new drug heralds 'momentous' advance in stroke prevention". Montreal Gazette. 29 October 2010. https://montrealgazette.com/health/Approval+drug+heralds+momentous+advance+stroke+prevention/3739714/story.html.
- ↑ "Pradax (Dabigatran Etexilate) Gains Approval In Canada For Stroke Prevention In Atrial Fibrillation" Medical News Today. 28 October 2010.
- ↑ "Dabigatran versus warfarin in patients with atrial fibrillation". The New England Journal of Medicine 361 (12): 1139–1151. September 2009. doi:10.1056/NEJMoa0905561. PMID 19717844.
- ↑ "New oral anticoagulants in atrial fibrillation". European Heart Journal 29 (2): 155–165. January 2008. doi:10.1093/eurheartj/ehm575. PMID 18096568.
- ↑ "Boehringer wins first US OK in blood-thinner race". Reuters. 19 October 2010. https://www.reuters.com/article/boehringer-pradaxa-idUSN1916563620101019.
- ↑ "FDA approves Pradaxa to prevent stroke in people with atrial fibrillation" (Press release). U.S. Food and Drug Administration (FDA). 19 October 2010. Archived from the original on 20 October 2010.
- ↑ Shirley S. Wang (20 September 2010). "New Blood-Thinner Recommended by FDA Panel". The Wall Street Journal. https://blogs.wsj.com/health/2010/09/20/new-blood-thinner-recommended-by-fda-panel/.
- ↑ "Use of emerging oral anticoagulants in clinical practice: translating results from clinical trials to orthopedic and general surgical patient populations". Annals of Surgery 250 (2): 219–228. August 2009. doi:10.1097/SLA.0b013e3181ae6dbe. PMID 19638915.
- ↑ "2011 ACCF/AHA/HRS focused update on the management of patients with atrial fibrillation (update on Dabigatran): a report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines". Circulation 123 (10): 1144–1150. March 2011. doi:10.1161/CIR.0b013e31820f14c0. PMID 21321155.
- ↑ "FDA Drug Safety Communication: FDA study of Medicare patients finds risks lower for stroke and death but higher for gastrointestinal bleeding with Pradaxa (dabigatran) compared to warfarin". 21 June 2019. https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-study-medicare-patients-finds-risks-lower-stroke-and-death-higher.
- ↑ "Dabigatran: how the drug company withheld important analyses". BMJ 349: g4670. July 2014. doi:10.1136/bmj.g4670. PMID 25055829.
- ↑ "Dabigatran, bleeding, and the regulators". BMJ 349: g4517. July 2014. doi:10.1136/bmj.g4517. PMID 25056265.
- ↑ "$650 Million to Settle Blood Thinner Lawsuits". The New York Times. 28 May 2014. https://www.nytimes.com/2014/05/29/business/international/german-drug-company-to-pay-650-million-to-settle-blood-thinner-lawsuits.html.
 |

