Chemistry:Adenosine thiamine triphosphate
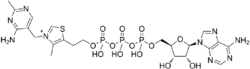
| |
| Names | |
|---|---|
| Systematic IUPAC name
(22R,23R,24S,25R)-16,134-Diamino-23,24,5,7,9-pentahydroxy-134,152-dimethyl-5,7,9-trioxo-4,6,8,10-tetraoxa-5λ5,7λ5,9λ5-triphospha-133λ5-1(9)-purina-15(5)-pyrimidina-13(5,3)-[1,3]thiazola-2(2,5)-oxolanapentadecaphan-133-ylium | |
| Other names
P1,P3-(Adenosine-5′-thiamine) triphosphate
| |
| Identifiers | |
| |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| MeSH | adenosine+thiamine+triphosphate |
PubChem CID
|
|
| UNII | |
| |
| |
| Properties | |
| C22H31N9O13P3S | |
| Molar mass | 754.52 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Adenosine thiamine triphosphate (AThTP), or thiaminylated adenosine triphosphate, is a natural thiamine adenine nucleotide.[1] It was discovered in Escherichia coli where it may account for up to 15 - 20% of total thiamine under carbon starvation. AThTP also exists in eukaryotic organisms such as yeast, roots of higher plants and animal tissues, albeit at a much lower concentration. It was found to exist in small amounts in the muscle, heart, brain, kidneys and liver of mice.[2]
In E. coli AThTP is synthesized from thiamine diphosphate (ThDP) according to the following reaction catalyzed by thiamine diphosphate adenylyl transferase:[3]
- ThDP + ATP (ADP) ↔ AThTP + PPi (Pi)
Structure and function
The molecule is made up of thiamine and adenosine joined together with phosphate groups. It is similar in structure to NAD+. The function of AThTP is not currently known but it has been shown to inhibit the activity of PARP-1.[2]
References
- ↑ "Discovery of a natural thiamine adenine nucleotide". Nat. Chem. Biol. 3 (4): 211–2. 2007. doi:10.1038/nchembio867. PMID 17334376.
- ↑ 2.0 2.1 "Adenosine thiamine triphosphate (AThTP) inhibits poly(ADP-ribose) polymerase-1(PARP-1) activity". J Nutr Sci Vitaminol (Tokyo) 57 (2): 192–6. 2011. doi:10.3177/jnsv.57.192. PMID 21697640.
- ↑ "Thiamine diphosphate adenylyl transferase from E. coli: functional characterization of the enzyme synthesizing adenosine thiamine triphosphate". BMC Biochem. 8: 17. 2007. doi:10.1186/1471-2091-8-17. PMID 17705845.
External links
- "A first for vitamins". Nature 446 (7132): 112–113. 2007. doi:10.1038/446112a.
- Jordan F (2007). "Adenosine triphosphate and thiamine cross paths". Nat. Chem. Biol. 3 (4): 202–3. doi:10.1038/nchembio0407-202. PMID 17372602.
 |

