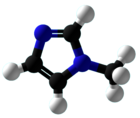
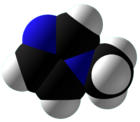
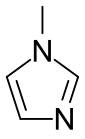
Chemistry:1-Methylimidazole
| |||
|
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
1-Methyl-1H-imidazole | |||
| Other names
1-Methylimidazole
N-Methylimidazole NMI | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| 105197 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
| EC Number |
| ||
| 2403 | |||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C4H6N2 | |||
| Molar mass | 82.10 g/mol | ||
| Density | 1.03 g/cm3 | ||
| Melting point | −6 °C (21 °F; 267 K) | ||
| Boiling point | 198 °C (388 °F; 471 K) | ||
| Hazards | |||
| Safety data sheet | Oxford MSDS | ||
| GHS pictograms |  
| ||
| GHS Signal word | Danger | ||
| H302, H312, H314 | |||
| P260, P264, P270, P280, P301+312, P301+330+331, P302+352, P303+361+353, P304+340, P305+351+338, P310, P312, P321, P322, P330, P363, P405, P501 | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
1-Methylimidazole or N-methylimidazole is an aromatic heterocyclic organic compound with the formula CH3C3H3N2. It is a colourless liquid that is used as a specialty solvent, a base, and as a precursor to some ionic liquids. It is a fundamental nitrogen heterocycle and as such mimics for various nucleoside bases as well as histidine and histamine.
Basicity
With the N-methyl group, this particular derivative of imidazole cannot tautomerize. It is slightly more basic than imidazole, as indicated by the pKa's of the conjugate acids of 7.0 and 7.4.[1] Methylation also provides a significantly lower melting point, which makes 1-methylimidazole a useful solvent.
Synthesis
1-Methylimidazole is prepared mainly by two routes industrially. The main one is acid-catalysed methylation of imidazole by methanol. The second method involves the Radziszewski reaction from glyoxal, formaldehyde, and a mixture of ammonia and methylamine.[2][3]
- (CHO)2 + CH2O + CH3NH2 + NH3 → H2C2N(NCH3)CH + 3 H2O
The compound can be synthesized on a laboratory scale by methylation of imidazole at the pyridine-like nitrogen and subsequent deprotonation.[4] Similarly, 1-methylimidazole may be synthesized by first deprotonating imidazole to form a sodium salt followed by methylation.[5][6]
- H2C2N(NH)CH + CH3I → [H2C2(NH)(NCH3)CH]I
- [H2C2(NH)(NCH3)CH]I + NaOH → H2C2N(NCH3)CH + H2O + NaI
Applications
In the research laboratory, 1-methylimidazole and related derivatives have been used as mimic aspects of diverse imidazole-based biomolecules.
1-Methylimidazole is also the precursor for the synthesis of the methylimidazole monomer of pyrrole-imidazole polyamides. These polymers can selectively bind specific sequences of double-stranded DNA by intercalating in a sequence dependent manner.[7]
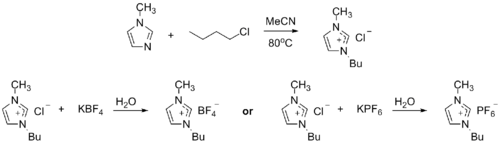
Ionic liquid precursor
1-Methylimidazole alkylates to form dialkyl imidazolium salts. Depending on the alkylating agent and the counteranion, various ionic liquids result, e.g. 1-butyl-3-methylimidazolium hexafluorophosphate ("BMIMPF6"):[8][9]
BASF has used 1-methylimidazole as a means to remove acid during their industrial-scale production of diethoxyphenylphosphine. In this biphasic acid scavenging using ionic liquids (BASIL) process, 1-methylimidazole reacts with HCl to produce 1-methylimidazolium hydrochloride, which spontaneously separates as a separate liquid phase under the reaction conditions.[8][10]
- 2 MeC3N2H3 + C6H5PCl2 + 2 C2H5OH → 2 [MeC3N2H4]Cl + C6H5P(OC2H5)2
Donor properties
1-methylimidazole (NMIz) as a ligand forms octahedral ions M(NMIz)62+with M = Fe, Co, Ni, and a square-planar ion Cu(NMIz)42+. [11] 1-methylimidazole forms adducts with Lewis acids such as molybdenum perfluorobutyrate and [Rh(CO)2Cl]2. The donor properties of 1-methylimidazole have been analyzed by the ECW model yielding EB= 1.16 and CB= 4.92.
See also
References
- ↑ Albert, A., Heterocyclic Chemistry, 2nd ed.; 1968 Athlone Press, ISBN:0-485-11092-X
- ↑ Ebel, K.; Koehler, H.; Gamer, A. O.; Jäckh, R. (2002). "Ullmann's Encyclopedia of Industrial Chemistry". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a13_661.
- ↑ Bronislaw Radziszewski (1882). "Ueber die Constitution des Lophins und verwandter Verbindungen" (in German). Berichte der deutschen chemischen Gesellschaft 15 (2): 1493–1496. doi:10.1002/cber.18820150207. https://zenodo.org/record/1425266.
- ↑ Gilchrist, T. L., Heterocyclic Chemistry, 2nd ed.; 1992 Longman Scientific & Technical, ISBN:0-582-06420-1
- ↑ Grimmett, M. R., Imidazole and Benzimidazole Synthesis; 1997 Academic Press, ISBN:0-12-303190-7
- ↑ Gupta, R. R., Kumar, M., Gupta, V., Heterocyclic Chemistry II: Five Membered Heterocycles; 1999 Springer, ISBN:3-540-65252-3
- ↑ Baird, Eldon E.; Dervan, Peter B. (1996). "Solid Phase Synthesis of Polyamides Containing Imidazole and Pyrrole Amino Acids". Journal of the American Chemical Society 118 (26): 6141–6. doi:10.1021/ja960720z. https://authors.library.caltech.edu/66889/2/ja6141.pdf.
- ↑ 8.0 8.1 Meindersma, G. Wytze; Maase, Matthias; De Haan, André B. (2007). "Ionic Liquids". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.l14_l01. ISBN 978-3-527-30673-2.
- ↑ Dupont, J.; Consorti, C.; Suarez, P.; de Souza, R. (2002). "Preparation of 1-Butyl-3-methyl imidazolium-based Room Temperature Ionic Liquids". Organic Syntheses 79: 236. doi:10.15227/orgsyn.079.023.
- ↑ Welton, Tom (11 November 2015). "Solvents and sustainable chemistry". Proceedings of the Royal Society A: Mathematical, Physical and Engineering Sciences 471 (2183): 20150502. doi:10.1098/rspa.2015.0502. PMID 26730217. Bibcode: 2015RSPSA.47150502W.

- ↑ Reedijk,R. (1969). "Pyrazoles and imidazoles as ligands. II. Coordination compounds of N-methyl imidazole with metal perchlorates and tetrafluoroborates". Inorganica Chimica Acta 3: 517–522. doi:10.1016/S0020-1693(00)92544-1.
 |