Chemistry:Heterocyclic compound
A heterocyclic compound or ring structure is a cyclic compound that has atoms of at least two different elements as members of its ring(s).[1] Heterocyclic organic chemistry is the branch of organic chemistry dealing with the synthesis, properties, and applications of organic heterocycles.[2]
Examples of heterocyclic compounds include all of the nucleic acids, the majority of drugs, most biomass (cellulose and related materials), and many natural and synthetic dyes. More than half of known compounds are heterocycles.[3] 59% of US FDA-approved drugs contain nitrogen heterocycles.[4]
Classification
The study of organic heterocyclic chemistry focuses especially on organic unsaturated derivatives, and the preponderance of work and applications involves unstrained organic 5- and 6-membered rings. Included are pyridine, thiophene, pyrrole, and furan. Another large class of organic heterocycles refers to those fused to benzene rings. For example, the fused benzene derivatives of pyridine, thiophene, pyrrole, and furan are quinoline, benzothiophene, indole, and benzofuran, respectively. The fusion of two benzene rings gives rise to a third large family of organic compounds. Analogs of the previously mentioned heterocycles for this third family of compounds are acridine, dibenzothiophene, carbazole, and dibenzofuran, respectively.
Heterocyclic organic compounds can be usefully classified based on their electronic structure. The saturated organic heterocycles behave like the acyclic derivatives. Thus, piperidine and tetrahydrofuran are conventional amines and ethers, with modified steric profiles. Therefore, the study of organic heterocyclic chemistry focuses on organic unsaturated rings.
Inorganic rings
Some heterocycles contain no carbon. Examples are borazine (B3N3 ring), hexachlorophosphazenes (P3N3 rings), and tetrasulfur tetranitride S4N4. In comparison with organic heterocycles, which have numerous commercial applications, inorganic ring systems are mainly of theoretical interest. IUPAC recommends the Hantzsch-Widman nomenclature for naming heterocyclic compounds.[5]
Notes on lists
- "Heteroatoms" are atoms in the ring other than carbon atoms.
- Some of the names refer to classes of compounds rather than individual compounds.
- Also no attempt is made to list isomers.
3-membered rings
Although subject to ring strain, 3-membered heterocyclic rings are well characterized.[6]
Three-membered rings with one heteroatom
| Heteroatom | Saturated | Unsaturated |
|---|---|---|
| Boron | Borirane | Borirene |
| Nitrogen | Aziridine | Azirine |
| Oxygen | Oxirane (ethylene oxide, epoxides) | Oxirene |
| Phosphorus | Phosphirane | Phosphirene |
| Sulfur | Thiirane (episulfides) | Thiirene |
Three-membered rings with two heteroatoms
| Heteroatoms | Saturated | Unsaturated |
|---|---|---|
| 2× Nitrogen | Diaziridine | Diazirine |
| Nitrogen + oxygen | Oxaziridine | Oxazirine |
| 2× Oxygen | Dioxirane (highly unstable) |
4-membered rings
Four-membered rings with one heteroatom
| Heteroatom | Saturated | Unsaturated |
|---|---|---|
| Nitrogen | Azetidine | Azete |
| Oxygen | Oxetane | Oxete |
| Phosphorus | Phosphetane | Phosphete |
| Sulfur | Thietane | Thiete |
Four-membered rings with two heteroatoms
| Heteroatoms | Saturated | Unsaturated |
|---|---|---|
| 2× Nitrogen | Diazetidine | Diazete |
| 2× Oxygen | Dioxetane | Dioxete |
| 2× Sulfur | Dithietane | Dithiete |
5-membered rings
Five-membered rings with one heteroatom
| Heteroatom | Saturated | Unsaturated |
|---|---|---|
| Antimony | Stibolane | Stibole |
| Arsenic | Arsolane | Arsole |
| Bismuth | Bismolane | Bismole |
| Boron | Borolane | Borole |
| Nitrogen | Pyrrolidine ("Azolidine" is not used) | Pyrrole ("Azole" is not used) |
| Oxygen | Tetrahydrofuran | Furan |
| Phosphorus | Phospholane | Phosphole |
| Selenium | Selenolane | Selenophene |
| Silicon | Silacyclopentane | Silole |
| Sulfur | Tetrahydrothiophene | Thiophene |
| Tellurium | Tellurophene | |
| Tin | Stannolane | Stannole |
Five-membered rings with two heteroatoms
The 5-membered ring compounds containing two heteroatoms, at least one of which is nitrogen, are collectively called the azoles. Thiazoles and isothiazoles contain a sulfur and a nitrogen atom in the ring. Dithiolanes have two sulfur atoms.
| Heteroatoms | Saturated | Unsaturated (and partially unsaturated) |
|---|---|---|
| 2× nitrogen | Imidazolidine Pyrazolidine |
Imidazole (Imidazoline) Pyrazole (Pyrazoline) |
| Oxygen + sulfur | 1,3-Oxathiolane 1,2-Oxathiolane |
Oxathiole (Oxathioline) Isoxathiole |
| Nitrogen + Oxygen | Oxazolidine Isoxazolidine |
Oxazole (Oxazoline) Isoxazole |
| Nitrogen + sulfur | Thiazolidine Isothiazolidine |
Thiazole (Thiazoline) Isothiazole |
| 2× oxygen | Dioxolane | |
| 2× sulfur | Dithiolane | Dithiole |
Five-membered rings with at least three heteroatoms
A large group of 5-membered ring compounds with three or more heteroatoms also exists. One example is the class of dithiazoles, which contain two sulfur atoms and one nitrogen atom.
| Heteroatoms | Saturated | Unsaturated |
|---|---|---|
| N N N | Triazoles | |
| N N O | Furazan Oxadiazole | |
| N N S | Thiadiazole | |
| N O O | Dioxazole | |
| N S S | Dithiazole | |
| N N N N | Tetrazole | |
| N N N N O | Oxatetrazole | |
| N N N N S | Thiatetrazole | |
| N N N N N | Pentazole |
6-membered rings
Six-membered rings with one heteroatom
| Heteroatom | Saturated | Unsaturated | Ions |
|---|---|---|---|
| Antimony | Stibinin[7] | ||
| Arsenic | Arsinane | Arsinine | |
| Bismuth | Bismin[8] | ||
| Boron | Borinane | Borinine | Boratabenzene anion |
| Germanium | Germinane | Germine | |
| Nitrogen | Piperidine (Azinane is not used) |
Pyridine (Azine is not used) |
Pyridinium cation |
| Oxygen | Oxane | Pyran (2H-Oxine is not used) |
Pyrylium cation |
| Phosphorus | Phosphinane | Phosphinine | |
| Selenium | Selenane | Selenopyran[9] | Selenopyrylium cation |
| Silicon | Silinane | Siline | |
| Sulfur | Thiane | Thiopyran (2H-Thiine is not used) |
Thiopyrylium cation |
| Tellurium | Tellurane | Telluropyran | Telluropyrylium cation |
| Tin | Stanninane | Stannine |
Six-membered rings with two heteroatoms
| Heteroatom | Saturated | Unsaturated |
|---|---|---|
| Nitrogen / nitrogen | Diazinane | Diazine |
| Oxygen / nitrogen | Morpholine | Oxazine |
| Sulfur / nitrogen | Thiomorpholine | Thiazine |
| Oxygen / Sulfur | Oxathiane | Oxathiin |
| Oxygen / oxygen | Dioxane | Dioxine |
| Sulfur / sulfur | Dithiane | Dithiin |
| Boron / nitrogen | 1,2-Dihydro-1,2-azaborine |
Six-membered rings with three heteroatoms
| Heteroatom | Saturated | Unsaturated |
|---|---|---|
| Nitrogen | Triazinane | Triazine |
| Oxygen | Trioxane | |
| Sulfur | Trithiane |
Six-membered rings with four heteroatoms
| Heteroatom | Saturated | Unsaturated |
|---|---|---|
| Nitrogen | Tetrazine |
Carborazine is a six-membered ring with two nitrogen heteroatoms and two boron heteroatom.
Six-membered rings with five heteroatoms
| Heteroatom | Saturated | Unsaturated |
|---|---|---|
| Nitrogen | Pentazine |
Six-membered rings with six heteroatoms
The hypothetical chemical compound with six nitrogen heteroatoms would be hexazine.
Borazine is a six-membered ring with three nitrogen heteroatoms and three boron heteroatoms.
7-membered rings
In a 7-membered ring, the heteroatom must be able to provide an empty π-orbital (e.g. boron) for "normal" aromatic stabilization to be available; otherwise, homoaromaticity may be possible. Compounds with one heteroatom include:
| Heteroatom | Saturated | Unsaturated |
|---|---|---|
| Boron | Borepin | |
| Nitrogen | Azepane | Azepine |
| Oxygen | Oxepane | Oxepine |
| Sulfur | Thiepane | Thiepine |
Those with two heteroatoms include:
| Heteroatom | Saturated | Unsaturated |
|---|---|---|
| Nitrogen | Diazepane | Diazepine |
| Nitrogen/sulfur | Thiazepine |
8-membered rings
| Heteroatom | Saturated | Unsaturated |
|---|---|---|
| Nitrogen | Azocane | Azocine |
| Oxygen | Oxocane | Oxocine |
| Sulfur | Thiocane | Thiocine |
Borazocine is an eight-membered ring with four nitrogen heteroatoms and four boron heteroatoms.
9-membered rings
| Heteroatom | Saturated | Unsaturated |
|---|---|---|
| Nitrogen | Azonane | Azonine |
| Oxygen | Oxonane | Oxonine |
| Sulfur | Thionane | Thionine |
Images of rings with one heteroatom
- Names in italics are retained by IUPAC and they do not follow the Hantzsch-Widman nomenclature
| Saturated | Unsaturated | ||||||
|---|---|---|---|---|---|---|---|
| Heteroatom | Nitrogen | Oxygen | Sulfur | Nitrogen | Oxygen | Sulfur | |
| 3-atom ring | Aziridine | Oxirane | Thiirane | Azirine | Oxirene | Thiirene | |
 |
Structure of oxirane | Structure of thiirane | Structure of azirine | Structure of oxirene | 
| ||
| 4-atom ring | Azetidine | Oxetane | Thietane | Azete | Oxete | Thiete | |
 |
Structure of oxetane | Structure of thietane | Structure of azete | Structure of oxete | 
| ||
| 5-atom ring | Pyrrolidine | Oxolane | Thiolane | Pyrrole | Furan | Thiophene | |
 |
Structure of oxolane | 
|
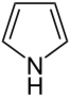 |
Structure of furan | 
| ||
| 6-atom ring | Piperidine | Oxane | Thiane | Pyridine | Pyran | Thiopyran | |
 |
Structure of oxane | 
|
 |
Structure of pyran | 
| ||
| 7-atom ring | Azepane | Oxepane | Thiepane | Azepine | Oxepine | Thiepine | |
 |
Structure of oxepane | Structure of thiepane | Structure of azepine | Structure of oxepine | 
| ||
| 8-atom ring | Azocane | Oxocane | Thiocane | Azocine | Oxocine | Thiocine | |
 |
Structure of oxocane | Structure of thiocane | Structure of azocine | Structure of oxocine | 
| ||
| 9-atom ring | Azonane | Oxonane | Thionane | Azonine | Oxonine | Thionine | |
 |
Structure of oxonane | Structure of thionane | Structure of azonine | Structure of oxonine | 
| ||
Fused/condensed rings
Heterocyclic rings systems that are formally derived by fusion with other rings, either carbocyclic or heterocyclic, have a variety of common and systematic names. For example, with the benzo-fused unsaturated nitrogen heterocycles, pyrrole provides indole or isoindole depending on the orientation. The pyridine analog is quinoline or isoquinoline. For azepine, benzazepine is the preferred name. Likewise, the compounds with two benzene rings fused to the central heterocycle are carbazole, acridine, and dibenzoazepine. Thienothiophene are the fusion of two thiophene rings. Phosphaphenalenes are a tricyclic phosphorus-containing heterocyclic system derived from the carbocycle phenalene.
History of heterocyclic chemistry
The history of heterocyclic chemistry began in the 1800s, in step with the development of organic chemistry. Some noteworthy developments:[10]
- 1818: Brugnatelli makes alloxan from uric acid
- 1832: Dobereiner produces furfural (a furan) by treating starch with sulfuric acid
- 1834: Runge obtains pyrrole ("fiery oil") by dry distillation of bones
- 1906: Friedlander synthesizes indigo dye, allowing synthetic chemistry to displace a large agricultural industry
- 1936: Treibs isolates chlorophyll derivatives from crude oil, explaining the biological origin of petroleum.
- 1951: Chargaff's rules are described, highlighting the role of heterocyclic compounds (purines and pyrimidines) in the genetic code.
Uses
Heterocyclic compounds are pervasive in many areas of life sciences and technology.[2] Many drugs are heterocyclic compounds.[11]
References
- ↑ IUPAC Gold Book heterocyclic compounds
- ↑ 2.0 2.1 Thomas L. Gilchrist "Heterocyclic Chemistry" 3rd ed. Addison Wesley: Essex, England, 1997. 414 pp. ISBN:0-582-27843-0.
- ↑ Rees, Charles W. (1992). "Polysulfur-Nitrogen Heterocyclic Chemistry". Journal of Heterocyclic Chemistry 29 (3): 639–651. doi:10.1002/jhet.5570290306.
- ↑ Edon Vitaku, David T. Smith, Jon T. Njardarson (2014). "Analysis of the Structural Diversity, Substitution Patterns, and Frequency of Nitrogen Heterocycles among U.S. FDA Approved Pharmaceuticals". J. Med. Chem. 57 (24): 10257–10274. doi:10.1021/jm501100b. PMID 25255204.
- ↑ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "Hantzsch–Widman name". doi:10.1351/goldbook.H02737
- ↑ Smith, Michael B.; March, Jerry (2007), Advanced Organic Chemistry: Reactions, Mechanisms, and Structure (6th ed.), New York: Wiley-Interscience, ISBN 978-0-471-72091-1, https://books.google.com/books?id=JDR-nZpojeEC&printsec=frontcover
- ↑ "Stibinin". Royal Society of Chemistry. http://www.chemspider.com/Chemical-Structure.119914.html.
- ↑ "Bismin". Royal Society of Chemistry. http://www.chemspider.com/Chemical-Structure.119910.html?rid=d49ac864-bf96-45d3-b78e-c54e585efbc4.
- ↑ "Selenopyranium". Royal Society of Chemistry. http://www.chemspider.com/Chemical-Structure.10431325.html?rid=15953636-c58f-4e8d-b945-9912511e750a.
- ↑ Campaigne, E. (1986). "Adrien Albert and the rationalization of heterocyclic chemistry". Journal of Chemical Education 63 (10): 860. doi:10.1021/ed063p860. Bibcode: 1986JChEd..63..860C.
- ↑ "IPEXL.com Multilingual Patent Search, Patent Ranking". http://www.ipexl.com/share/f593e64bc3679cb669d02c007efdca17.
External links
- Hantzsch-Widman nomenclature, IUPAC
- Heterocyclic amines in cooked meat, US CDC
- List of known and probable carcinogens, American Cancer Society
- List of known carcinogens by the State of California, Proposition 65 (more comprehensive)
 |



